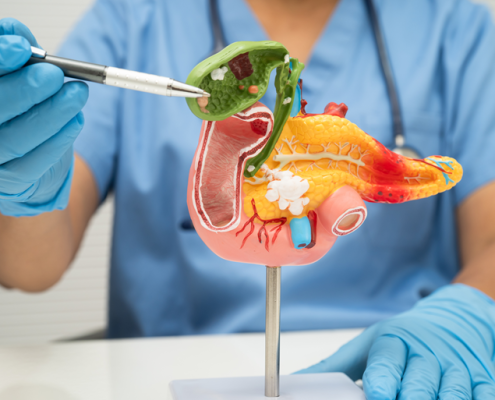
ICRC Scientists Join Forces with South Korea to Tackle Deadly Pancreatic Cancer
ICRC, a joint workplace of St. Anne’s University Hospital in Brno and the Faculty of Medicine at Masaryk University, is launching an international research collaboration with Gyeongsang National University in South Korea. The project will…

CLARA Young Innovator Award 2026: The 1st Edition Focuses on Population Ageing
The CLARA Young Innovator Award 2026 has been officially announced,inviting young researchers, developers, and innovators under 35 years of age to present digital solutions addressing the challenges and opportunities of ageing societies. Apply…

ICRC Scientists Develop AI Tool to Help Cardiologists Treat Atrial Fibrillation, Potentially Cutting Surgeries by One Third
Around half a million people in the Czech Republic suffer from one of the most common heart arrhythmias, atrial fibrillation. A research project led by Jakub Hejč at ICRC focuses on personalizing treatment and reducing the likelihood of recurrence.…

